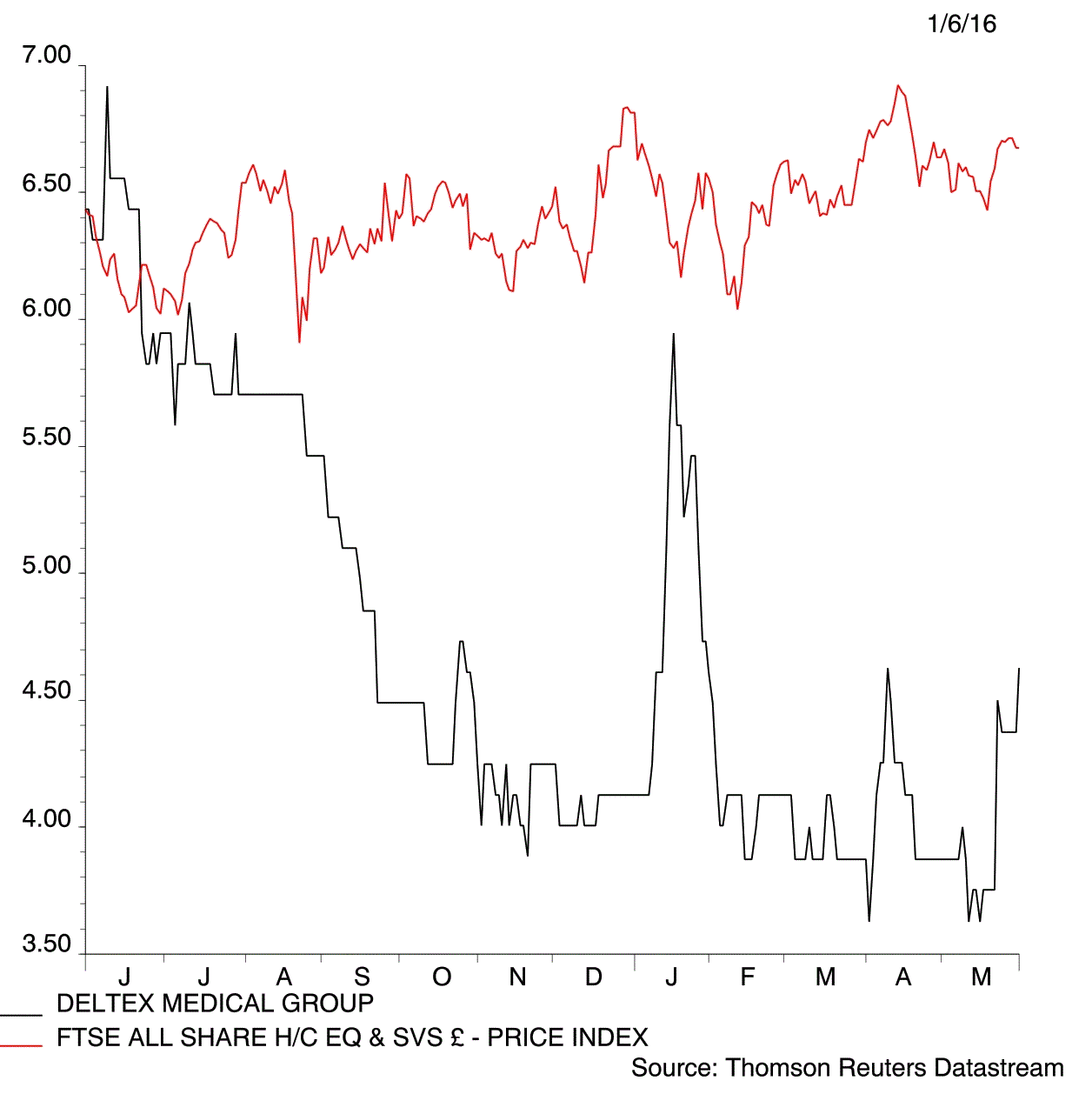
The power a positive clinical trial result has on the market was evident at blood monitor-maker Deltex Medical (DEMG:AIM) on Wednesday.
Shares climbed 5.7% to 4.6p on initial tests showing that its oesophageal Doppler blood flow monitor eliminates almost three quarters of the complications that can follow surgery.
The tests reduced post-operative complications from 198 to 56, or by 72%.
The Doppler device monitors blood flow in real time to ensure that fluids are reaching every part of the body, which can speed up the recovery of those undergoing surgery or are in intensive care.
The randomised trial involved 450 patients in five hospitals in Spain who underwent gastrointestinal, urological, gynaecological and orthopaedic surgery.
Deltex intends to expand its business in Spain following the tests.
Fewer problems following an operation means lower costs, a selling point Deltex’s management are keen to push to cash-strapped health services.
The results also showed that the device could reduce a patient’s hospital stay by two days, another cost-cutting benefit.